Experimental Technique
Objects were measured using a Vernier Caliper and a digital mass balance.
Data
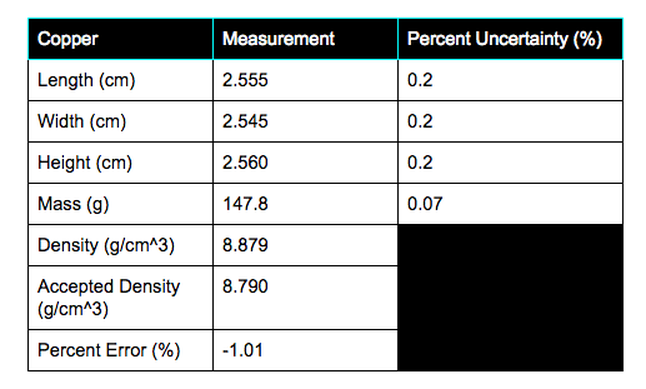
All measurements and results are organized into three data charts.
Analysis
These are sample calculations from the physics lab.
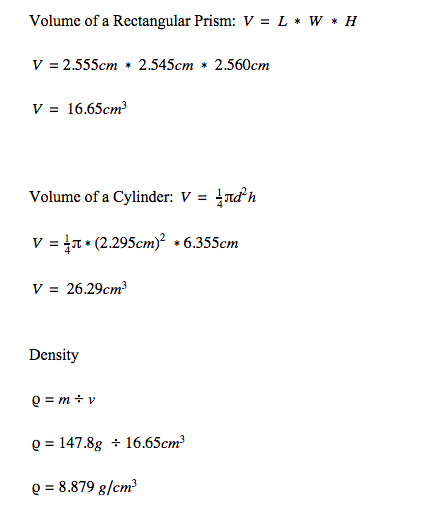
Conclusion
The percent error for the density measurement was below 3% for all measurements. Copper was measured to be 8.879 g/cm^3, while the accepted value was 8.790 g/cm^3. Therefore, copper's percent error is 1.01%. Acrylic was measured to be 1.15 g/cm^3, while the accepted value was 1.185 g/cm^3. Therefore, acrylic's percent error is 2.95%. Polypropylene was measured to be 0.893 g/cm^3, while the accepted value was 0.9 g/cm^3. Therefore, polypropylene's percent error is 0.8%. One source of error is the rounded edges of the acrylic object. It was assumed to be a cube, but was actually a rectangular prism. Another error is the fact that the balance only measures to the tenth gram. To fix this error, we could find a balance that measures out to more decimal places for a more precise measurement.